Many people from outside of the realm of academia (or is it a fiefdom?) prefer to get the latest scoops on new paleontological or geological research directly from the source, rather than just reading a press release or news article about it. As someone looking from the inside out, I’m pleased to see so many non-scientists try to probe one layer deeper with their understanding of a beloved scientific topic that interests them, and I try to encourage it through my own blogging, speaking, teaching, and other forms of outreach.
 An alligator den on St. Catherines Island, (Georgia), with baby alligator and “big momma” alligator for scale. This week, I presented a poster with about these big burrows and their makers at the Society of Vertebrate Paleontology meeting in Raleigh, North Carolina. The original field work we did for this research was reported back in March here, and now we’re ready to share more of what we found out. (Photograph by Anthony Martin.)
An alligator den on St. Catherines Island, (Georgia), with baby alligator and “big momma” alligator for scale. This week, I presented a poster with about these big burrows and their makers at the Society of Vertebrate Paleontology meeting in Raleigh, North Carolina. The original field work we did for this research was reported back in March here, and now we’re ready to share more of what we found out. (Photograph by Anthony Martin.)
Unfortunately, many of the original research articles that become subjects of media attention are behind paywalls, requiring a reader to pay for access to read those articles, even if the research was publicly funded. This practice is especially common if the research is published in one of those glamorous journals that seemingly make or break academic careers in science, regardless of the lasting quality of the research. (I won’t name them directly, but let’s just say that’s the nature of science nowadays.)
So one option for these curious folks is to read abstracts from proceedings volumes of professional meetings. Abstracts, which ideally are succinct summaries highlighting the most significant findings of a given study, can thus serve as a way for the public to at least get a few insights on the latest scientific research happening in their favorite disciplines.
 Want to get below the surface with this research? Oh, sorry, I was just being metaphorical. You really don’t want to go below the surface of an alligator den, which is why we mostly studied abandoned ones, mapped them, and otherwise tried to use methods that didn’t bother the alligators or otherwise have uncomfortable encounters with them.
Want to get below the surface with this research? Oh, sorry, I was just being metaphorical. You really don’t want to go below the surface of an alligator den, which is why we mostly studied abandoned ones, mapped them, and otherwise tried to use methods that didn’t bother the alligators or otherwise have uncomfortable encounters with them.
Along those lines, the annual meeting of the Society of Vertebrate Paleontology (SVP) has been taking place this week in Raleigh, North Carolina, and it has an abstract volume associated with the meeting. Regrettably, though, the general public does not have access to these abstracts, only SVP members and people who have registered for the meeting. The Society of Vertebrate Paleontology also has a policy regarding researchers who publicly share their research results based on these abstracts, muddied by the word “embargo.” In short, this policy holds that people working for the media, which include reporters and bloggers (the latter of whom are also sometimes reporters), cannot write about and otherwise publicize research results presented at the meeting. That is, unless the researchers have given their permission to do so, or the results have been freely distributed by the researchers through a press release, blog, or other forms of outreach.
So in the spirit of the public having easier access to this primary scientific information, the following is our SVP abstract, which I presented as a poster at the meeting yesterday. The abstract is co-authored with Michael Page (Emory University), Sheldon Skaggs (Georgia Southern University), and R. Kelly Vance (also Georgia Southern University), and we worked together on the research, writing, and editing of the abstract. Because this abstract also includes a lot of scientific shorthand (charitably referred to as “jargon”), I also included a sentence-by-sentence explanation of it, in which the abstract text is in italics and my explanation is in formal typeface. So I hope you, the gentle reader, get something from this exercise in explanation, and we look forward to sharing more of this research with you as it continues to evolve and we publish it sometime next year as a peer-reviewed paper.
DENS OF THE AMERICAN ALLIGATOR (ALLIGATOR MISSISSIPPIENSIS) AS TRACES AND THEIR PREDICTIVE VALUE FOR FINDING LARGE ARCHOSAUR BURROWS IN THE GEOLOGIC RECORD
MARTIN, Anthony J., Emory University, Atlanta, GA, United States; PAGE, Michael, Emory University, Atlanta, GA, United States; SKAGGS, Sheldon, Georgia Southern University, Statesboro, GA, United States; VANCE, Robert K., Georgia Southern University, Statesboro, GA, United States
Large archosaur burrows are rarely interpreted from the geologic record, a circumstance that may be attributable to a lack of search images based on modern examples, rather than actual rarity.
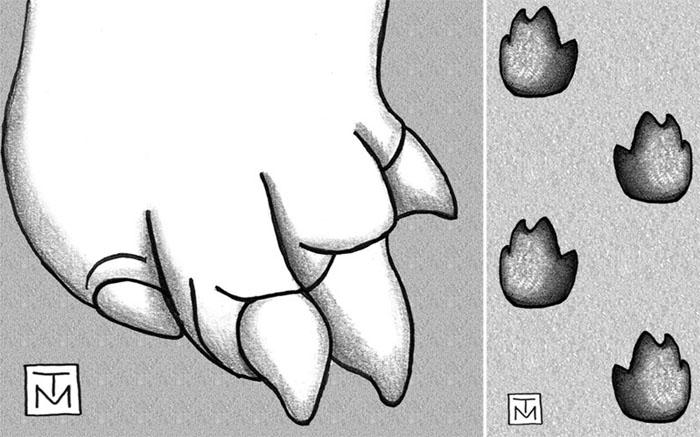
Archosaurs make up an evolutionarily related group of vertebrates that include crocodilians (alligators and crocodiles), dinosaurs (the non-bird ones, that is), birds, and their extinct relatives. A few of the larger extinct archosaurs may have dug burrows, but paleontologists have reported very few of these, with one exception being the small Cretaceous ornithopod dinosaur Oryctodromeus cubicularis, found in its burrow with two juveniles of the same species. The authors are proposing here that this “rarity” of archosaur burrows in the fossil record might be more attributable to paleontologists not knowing what modern archosaur burrows look like. So they don’t recognize the fossil ones, leading to a perceived rarity rather than an actual one.
To test this idea, we measured, imaged, and mapped den structures of the American alligator (Alligator mississippiensis) on St. Catherines Island (Georgia, USA).
By “measured,” I mean that my colleagues and I used a low-tech instrument known as a “tape measure” to assess the width and height of an alligator den entrance. By “imaged,” we used a much more technologically complex instruments and method, called ground-penetrating radar (GPR) in combination with computers to figure out what these dens looked like below the surface. By “mapped,” I mean that we looked for alligator dens on St. Catherines Island (Georgia) and recorded their locations using a handheld GPS (global positioning system) unit, then plotted the distribution of these points to see if any patterns emerged.
St. Catherines is an undeveloped barrier island on the Georgia coast, consisting of Pleistocene and Holocene sediments.
St. Catherines Island is undeveloped in the sense that very few buildings or people live on the island year-round. It is privately owned and reserved for researchers’ uses under the direction of the St. Catherines Island Foundation. Like most of the Georgia barrier islands on the southern part of its coast, St. Catherines also has a geologically complex history. Its northwestern end is made of sediments deposited about 40,000 years ago – during the Pleistocene Epoch – whereas its southeastern end is made of much more recent sediments from the Holocene Epoch.
Alligators dug most dens along the edges of freshwater ponds in loosely consolidated Holocene or Pleistocene sand.
This sentence doesn’t need much more explanation other than to reemphasize that alligators gravitate to freshwater ecosystems to dig their dens (pictured below), not saltwater ecosystems, like salt marshes or coastal dunes.
 Adult female alligators use dens to protect offspring, but burrows also aid in thermoregulation or serve as refugia for alligators during droughts and fires.
Adult female alligators use dens to protect offspring, but burrows also aid in thermoregulation or serve as refugia for alligators during droughts and fires.
This is probably the neatest insight we gained from doing the research, is that the dens aren’t just used by big momma ‘gators for raising baby ‘gators, but also to make sure alligators of all ages are cozy during winters, stay wet during droughts, and are safe from fires. For instance, because southern Georgia has been going through a drought the past few years, some of the occupied dens we saw were in places that were high-and-dry, but the dens themselves intersected the local water table (seen in one photo above).
Some dens are evidently reused and modified by different alligators after initial construction.
This is an important point for paleontologists to know, and probably shouldn’t have been buried so far into the abstract, but we couldn’t very well put it at the beginning, either. Dens, like other homes, get used again, and probably by generations of alligators. This means that once a den is dug, stays open, and has a wetland nearby, alligators may just move into an abandoned den and modify it if needed, an alligator form of “home improvement.”
Drought conditions along the Georgia coast have exposed many abandoned dens, thus better allowing for their study while increasing researcher safety.
The drought is bad for alligators but was good for us when we did our field work, because so many dens were abandoned and exposed on dry land. This also eased any concerns we had about bothering the alligators, but especially alleviated worries we might have had about close encounters with protective parents near occupied dens. To be sure, we ran into a few of those, but not as many as we would have if conditions had been wetter.
 Den entrances have half-moon cross-sections, and based on one sample (n = 20), these range from 22-115 cm wide (mean = 63 + 23 cm) and 14-55 cm high (23 + 9 cm).
Den entrances have half-moon cross-sections, and based on one sample (n = 20), these range from 22-115 cm wide (mean = 63 + 23 cm) and 14-55 cm high (23 + 9 cm).
I like throwing numbers into ichnology, just to remind people that this is a part of it as a science. Although our sample size is small compared to other studies of traces and trace fossils, it gives people an idea of the range of sizes of these dens, or at least their entrances. As an exercise in the imagination, think about whether you could squeeze into one of these. You know, if you were crazy enough to do such a thing.
In addition to field descriptions, we applied geographic information systems (GIS) and ground-penetrating radar (GPR) to help define the ecological context and subsurface geometry of these structures, respectively.
Computer-aided mapping methods like GIS helped us to test how alligators decided to make dens as a function of the landscape. For instance, we found most of their dens were in lower-elevation areas, which made sense when you think about water accumulating in those places. And the GPR served the dual purpose of not bothering the alligators if they were in their dens, while also keeping us away from their, um, denizens. (Sorry.)
GIS gave spatial data relatable to alligator territoriality, substrate conditions, and proximity to potential nest sites. GPR produced subsurface images of active dens, which were compared to abandoned dens for a sense of taphonomic history.
Big alligators tend to stay away from other big alligators. They also tend to burrow in sediments that don’t take too much effort for them. Female alligators also make their nests close to water bodies and dens, so their little tykes don’t have to travel so far to the water. Newer, active dens were also compared to those no longer being used to see what happens to them over time with neglect, kind of like how an old, abandoned house tends to fall apart and collapse on itself over time.
Most den entrances are southerly facing, with tunnels dipping to the northwest or northeast.
This is pretty self-explanatory, but I’ll just ask readers to think about why these dens are oriented like this.
From entrances, tunnels slope at about 10-15°, turn right or left within a meter, and lead to enlarged turn-around chambers.
Pure description here too, but by “turn-around chamber,” that means the den has enough room inside the den for a big adult alligator to go in head-first and turn around so that it’s head is right at the entrance. (See the photo of “big momma” at the top for an example of that.)
Collapsed dens in formerly ponded areas (secondary-succession maritime forests) provided further insights into subsurface forms of these structures.
Dens left high-and-dry from years ago and taken over by forests collapsed in a way that we could see the full outline of the den and measure these.
These features are: 3.1-4.6 m long; 30-40 cm deep, relatively narrow at either end (35-60 cm), and 1.2-1.6 m wide in their middles.
Dude. Those are big burrows. Dude.
Expansive areas were probable turn-around chambers, and total volumes of collapsed dens accordingly reflect maximum body sizes of their former occupants.
The bigger the den, the easier it was for a large occupant to turn around in it. And although smaller, younger alligators could have lived in these dens, some of the dens were too small to allow the really big alligators from moving into them.
One sampled area (8,100 m2), an almost dry former pond, had 30 abandoned dens, showing how multiple generations of alligators and fluctuating water levels can result in dense concentrations of alligator burrows over time.
Think of an area about the size of an American football field, and put 30 alligator dens in that area. (Now that would make for an interesting game, wouldn’t it?) These dens weren’t all made at the same time, though, and were constructed or abandoned as the pond filled or dried out, respectively.
In summary, the sheer abundance, distinctive traits, and sizes of these structures on St. Catherines and elsewhere in the Georgia barrier islands give paleontologists excellent search images for seeking similar trace fossils made by large semi-aquatic archosaurs.
That’s the big take-home message here for vertebrate paleontologists. All of the information we gathered about these alligator dens from the Georgia barrier islands, especially what they look like, can be applied to test the fossil record of archosaurs. In other words, did archosaurs actually leave lots of dens for us to find, but we just didn’t know what to look for? Hopefully we’ll find out because of this research.
 Later, denning ‘gator. (Photograph by Anthony Martin, taken on St. Catherines Island, Georgia.)
Later, denning ‘gator. (Photograph by Anthony Martin, taken on St. Catherines Island, Georgia.)
(Special thanks to Ruth Schowalter for assisting with the field work, and to the St. Catherines Island Foundation for funding some of the research.)
Source of Abstract (Reference):
Martin, A.J., Page, M., Vance, R.K., and Skaggs, S. 2012. Dens of the American alligator (Alligator mississippiensis) as traces and their predictive value for finding large archosaur burrows in the geologic record. Journal of Vertebrate Paleontology, 32 [Suppl. to No. 3]: 136.
 A seahorse, of course, is not a horse. But that’s not the only way seahorses differ from horses, in that they leave trails instead of tracks. Intrigued? Yeah, me too. (Photograph by Anthony Martin, taken at the UGA Aquarium, Skidaway Island, Georgia.)
A seahorse, of course, is not a horse. But that’s not the only way seahorses differ from horses, in that they leave trails instead of tracks. Intrigued? Yeah, me too. (Photograph by Anthony Martin, taken at the UGA Aquarium, Skidaway Island, Georgia.) A seahorse (Hippocampus sp.) showing off its lack of swimming skills by moving along the sandy bottom of a tank. Gee, what are all of those meandering and intersecting grooves in the sand and circular imprints? I wonder what made those? Sorry, first guess doesn’t count. (Photograph by Anthony Martin, taken at the UGA Aquarium, Skidaway Island, Georgia.)
A seahorse (Hippocampus sp.) showing off its lack of swimming skills by moving along the sandy bottom of a tank. Gee, what are all of those meandering and intersecting grooves in the sand and circular imprints? I wonder what made those? Sorry, first guess doesn’t count. (Photograph by Anthony Martin, taken at the UGA Aquarium, Skidaway Island, Georgia.) A seahorse making tail trails while swimming along the bottom of an aquarium. Notice how the trail would become less linear, wider, and more circular if the tail flops over to one side or another, involving a greater area of the curled end. (Photograph by Anthony Martin, taken at the UGA Aquarium, Skidaway Island, Georgia.)
A seahorse making tail trails while swimming along the bottom of an aquarium. Notice how the trail would become less linear, wider, and more circular if the tail flops over to one side or another, involving a greater area of the curled end. (Photograph by Anthony Martin, taken at the UGA Aquarium, Skidaway Island, Georgia.) A close-up of those trails left by swimming seahorses dragging their tails along a sandy surface. Also, check out the overlapping circular “plop” traces on the right, made by the curled part of the tail? (Photograph by Anthony Martin, taken at the UGA Aquarium, Skidaway Island, Georgia.)
A close-up of those trails left by swimming seahorses dragging their tails along a sandy surface. Also, check out the overlapping circular “plop” traces on the right, made by the curled part of the tail? (Photograph by Anthony Martin, taken at the UGA Aquarium, Skidaway Island, Georgia.)